Mechanisms Underlying Opioid Analgesia
Introduction to Opioids
Opioids are a class of drugs that are either derived from, or mimic the effects of natural substances (opiates) found in the opium poppy plant. Opioids exert their effects through binding to different subtypes of opioid receptors to produce a variety of effects including pain relief or analgesia.
Definitions
-
Acute Pain: Pain of sudden onset that is caused by something specific such as surgery, dental work, burns, cuts, or other body injuries. Acute pain goes away when the acute injury has healed.
-
Analgesia: The absence of pain.
-
Analgesic: A drug that reduces pain.
-
Chronic Pain: Pain that typically lasts for 6 months or more, and continues after an injury or illness has healed or been resolved. Chronic pain is often linked to conditions such as arthritis, cancer, back injuries or fibromyalgia.
-
Inflammatory Pain: Pain resulting from activation of nociceptive receptors by bioactive cytokines (e.g. ATP, bradykinin, prostanoids) released by activated cells of the immune system.
-
Narcotic: An American legal term derived from the Greek word meaning “numbness” (Anstie 1865)(https://www.merriam-webster.com/dictionary/narcotic). It can refer to over 116 drugs having psychoactive properties with legally restricted or prohibited use including opioids, cocaine, marijuana and LSD (International Narcotics Control Board 1995-2009).
-
Nociceptive Pain: The sensation of pain that results from damage to body tissues in response to external mechanical, thermal or chemical stimuli. Such sensations of pain serves an important physiological role as an alert mechanism to imminent danger.
-
Opiate: naturally occurring opioid agonists derived from opium, such as morphine & codeine (Figure 1).
-
Opioid: all compounds (natural or synthetic) that bind to opioid receptors. Examples include hydrocodone, oxycodone, fentanyl, and opioid receptor antagonists (e.g. naloxone & naltrexone).

-
Figure 1. Opium, opiates and opioids. Image has been reproduced from Wikipedia Commons.
Opioid Receptors
Opioid receptors are part of the G-protein-coupled receptor (GPCR) superfamily of receptors, and control different aspects of cellular function. The basic structure of GPCRs consist of a single polypeptide chain that crosses the cell membrane 7 times, resulting in alternate names of “7-transmembrane domain receptors” or “serpentine receptors” (Figure 1). Each GPCR has an extracellular N-terminal domain of variable length and an intracellular C-terminal domain that interacts with heterotrimeric G-proteins consisting of alpha, beta and gamma subunits (Saleem & Nisar, 2022; Dhaliwal & Gupta, 2023).
Opioid Receptor Signal Transduction
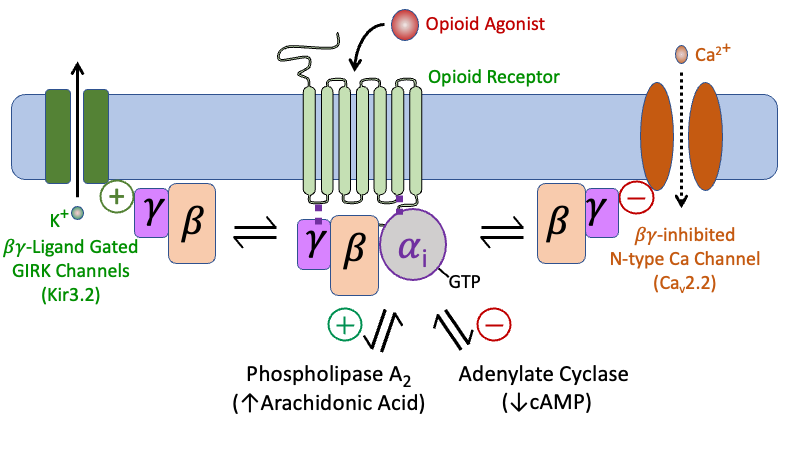
Figure 2. Opioid Signal Transduction involving Gi, and the most common molecular targets. Endogenous ligands (e.g. endorphins or enkephalins) or exogenous opioids (e.g. morphine, fentanyl) bind to opioid receptors and activate the Gi mediated signal transduction pathway, resulting in the exchange of GDP for GTP, and the separation of αi from βγ subunits). This can initiate multiple types of signal transduction including: i) βγ subunit inhibition of voltage-gated N-type (Cav2.2) Ca channels (Raingo et al 2007); ii) βγ subunit activation of G protein-gated inwardly rectifying potassium channels (GIRKs) (Huang et al, 1995); iii) αi inhibition of adenylate cyclase which will decrease cAMP and reduce the activity of cAMP-modulated HCN2 pacemaker channels (Ingram & Williams 1994); and iv) activation of phospholipase A2 that will increase the release of arachidonic acid, and increase the production of 12-HPETE by 12-LOX, resulting in the activation of voltage dependent K channels (Vaughan et al 1997; Christie et al 1999; Zhang et al, 2015).
Opioid Receptor Terminology
Multiple subtypes of opioid receptor subtypes have been identified that bind either exogenous or endogenous opioids. Most opioid analgesics exert their effects by binding to three opioid receptors subtypes known as the mu-opioid receptor (MOR), delta-opioid receptor (DOR) and kappa-opioid receptor (KOP) which are encoded by unique genes (Table 1) (Saleem & Nisar, 2022)(Table 1).
Table 1: Major Opioid Receptors
-
DOR: delta (δ) opioid receptor
-
Subtypes: DOR1, DOR2
-
Function: Analgesia, Convulsant, Physical dependence
-
G protein: Gi
-
-
KOR: kappa (κ) opioid receptor
-
Subtypes: KOR1, KOR2, KOR3
-
Function: Analgesia, Anticonvulsant, Depression, Dysphoria, Hallucination, Miosis, Stress
-
G protein: Gi
-
-
MOR: mu (μ) opioid receptor
-
Subtypes: MOR1, MOR2, MOR3
-
Function: MOR1: Analgesia, Physical dependence; MOR2: Euphoria, Respiratory depression, Miosis, Reduced GI motility, Physical dependence
-
G protein: Gi
-
Table 2: Opioid Related Receptors
-
NOR: nociceptin/orphanin FQ (N/OFQ) peptide receptor 1)
-
Function: integrating sensory processing, including pain transmission and modulation (Ding et al, 2023)
-
Ligands: nociceptin (a 17 amino acid neuropeptide). Has little or no affinity for morphine-like compounds or opioid peptides (Butour et al, 1997).
-
G proteins: Gi, Go & others
-
-
ZOR: zeta (ζ) receptor. Also known as the Opioid Growth Factor Receptor (OGFr)
-
Function: regulation of tissue growth in embryonic development, wound repair & in cancer (Zagon et al, 2002)
-
Ligand: met-enkephalin
-
-
MRGPRX2: Mas-Related G-Protein coupled Receptor member X2. Has been referred to as an “Atypical Opioid Receptor”.
-
Function: Not structurally related to opioid receptors. Expressed by cutaneous mast cells. Induces mast cell degranulation & a non-IgE mediated immune response when stimulated by a variety of chemically diverse positively charged molecules including vancomycin and the dextro-enantiomers of morphine, codeine & dextromethorphan 2). Does not bind fentanyl, naloxone or naltrexone.
-
The Pathophysiologic Basis for Pain
Pain is a distressing sensation caused by intense or damaging stimuli that serves a fundamentally important function to warn or signal that bodily harm has occurred or is imminent, and motivates a withdrawal from the damaging situation to protect the body.
The major components of pain sensation and transduction are summarized in Figure 3. Axons of primary afferents associated with nociceptive nerve endings produce action potentials in response to thermal, mechanical, or chemical stimuli that conduct sensory information from the site of injury in the periphery to the dorsal horn in the spinal column, which then transmit signals to secondary projection neurons that conduct pain signals to the brain stem and thalamus, which relay signals to the sensory cortex of central nervous system (CNS). Transmission of pain signals is also modulated at every level by inhibitory and excitatory interneurons (Bingham et al, 2009). Inflammatory pain (e.g. which can result from conditions such as gout or rheumatoid arthritis) utilizes the same pathway, but is initiated by the formation of inflammatory cytokines, as illustrated in Figure 4.
The Nociceptive Pathway & its Modulation
-
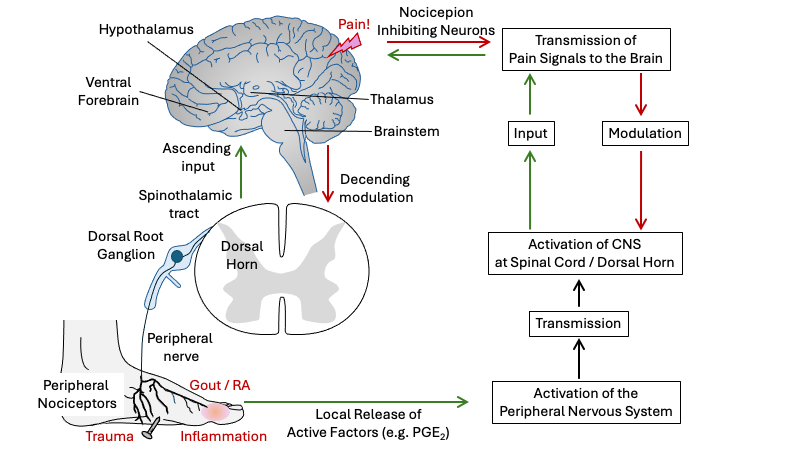
Figure 3. The pain pathway. Activation of peripheral pain receptors (nociceptors) by painful stimuli including inflammation-induced production of prostaglandins stimulates receptors on nociceptive nerve endings, resulting in an increase in the frequency of action potentials conducted to the dorsal horn of the spinal cord via the dorsal root ganglion. From the dorsal horn, the such pain signals are carried along the ascending pain pathway (spinothalamic tract) to the thalamus and cortex. Pain neurotransmission can be modulated by both pain-inhibiting and pain-facilitating neurons. Descending signals originating in supraspinal centers can modulate activity in the dorsal horn by controlling (inhibiting) the neurotransmission of ascending spinal pain signals. Abbreviations: CNS (Central Nervous System); PGE2 (Prostaglandin E2). Adapted from Bingham et al (2009).
Cellular Mechanisms Underlying Nociceptive & Inflammatory Pain
Cellular injury due to either trauma and/or inflammation will release intracellular substances into the extracellular space that have “damage-associated molecular patterns” (DAMPs) associated with them. These DAMPs are recognized by pattern recognition receptors on cells of the innate immune system, such as tissue resident mast cells and monocytes, triggering the release of inflammatory cytokines & chemotactic compounds that initiate an inflammatory response. Similarly, recognition of small molecules released by bacteria, viruses or fungi that contain “pathogen-associated molecular patterns” (PAMPs) are recognized as foreign and trigger a similar immune response (Wood & Goldberg, 2022).
There are numerous inflammatory mediators released by activated cells of the innate immune system including protons (acidic pH), kinins (e.g. bradykinin), prostanoids (e.g. PGE2), ATP, Serotonin (5-HT), histamine, leukotrienes (LTs), hydroxyeicosatetraenoic acid metabolites (HETEs) of arachidonic acid metabolism, and neuropeptides released by sensory nerve endings (e.g. CGRP, Substance P) (Dray, 1995; Wood & Goldberg, 2022). When these substances are release during the immune response in response to infection or trauma, they interact with receptors located on nociceptive nerve endings, resulting in membrane depolarization and triggering of trains of action potential by voltage sensitive Na channels (Nav1.8), which are then relayed by the peripheral afferent pain fibers to the spinal cord and pain processing centers in the central nervous system (Figure 4)(Prescott & Ratte, 2017; Chen et al 2013).
Mechanisms evoked by inflammatory mediators that result in increased depolarization and stimulation of nociceptor nerve terminals include:
-
Acidic pH & HETEs: activate TRPV1 (transient receptor potential cation channel member V1; known as the capsaicin receptor or the vanilloid receptor 1). TRPV1 is a nonselective cation channel that is also activated by heat (temperature >43oC / 109oF), acidic conditions, capsaicin and mustard oil; activation of TRPV1 results in membrane depolarization and action potential generation in sensory nerve endings (Benitez-Angeles et al, 2020).
-
PGE2 & Bradykinin: Prostaglandin E2 and bradykinin stimulate EP1 & B2 receptors which activate PKC via Gq, which increases channel phosphorylation and probability of channel opening for both TRPV1 and P2X3 cation-selective ion channels (Paulert et al, 2001; Wirkner et al, 2005; Brown & Yule, 2007). In addition, PGE2 activation of EP2 receptors activates adenylyl cyclase via Gs, resulting in an increase of cAMP, which activates HCN2 (the cation-selective pacemaker channel), a channel that has been identified as playing an important role in the neurogenesis of pain (Ingram & Williams, 1994; Emery et al, 2011 & 2012).
-
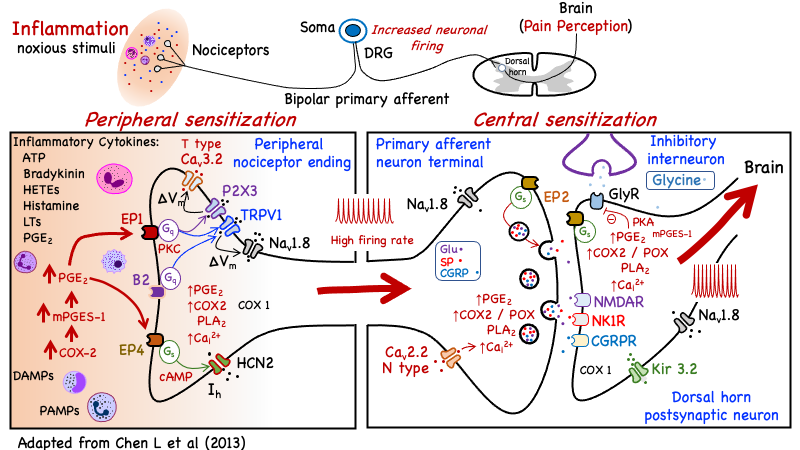
Figure 4. Cellular mechanisms underlying inflammatory pain signaling & molecular targets for analgesic drugs including NSAIDs, acetaminophen, opioids & suzetrigine (Nav1.8 inhibitor). Left Panel: Both COX-2 and Microsomal Prostaglandin E Synthase-1 (mPGES-1) undergo upregulation in both injured cells and infiltrating immune cells in inflamed tissues, which increases PGE2 levels at peripheral nociceptor nerve terminals. PGE2 contributes to peripheral sensitization by potentiation of TRPV1 and T-type (Cav3.2) calcium channel activity, as well as activation of TTX-resistant Na channels (Nav1.8) and purinergic P2X3 receptors channels mediated by prostanoid EP1 and EP4 receptors. Right Panel: Inflammation upregulates the expression of COX-2 and mPGES-1 in the spinal cord resulting in increased production of PGE2. In this region PGE2 mainly acts on EP2 receptors, which contributes to central sensitization through both pre- and post-synaptic mechanisms. At the presynaptic level, PGE2 facilitates the spinal release of different excitatory neurotransmitters glutamate (Glu), substance-P (SP), and calcitonin gene-related peptide (CGRP), resulting in enhanced nociceptive signaling. At the post-synaptic level, PGE2 directly enhances glutamate receptor (AMPAR and NMDAR) activity, and blocks inhibitory neurotransmission by glycine. Abbreviations: AMPAR (quisqualate receptor); B2 (bradykinin receptor); CGRPR (CGRP receptor); DAMPs (damage-associated molecular patterns); DRG (Dorsal Root Ganglion); HCN2 (Potassium/sodium hyperpolarization-activated cyclic nucleotide-gated ion channel 2); HETE (Hydroxyeicosatetraenoic acid); Nav1.8 (tetrodotoxin-resistant sodium channels); LTs (leukotrienes); NK1R (neurokinin-1 / substance P receptor); NMDAR (N-methyl-d-aspartic acid receptor); PAMPs (pathogen-associated molecular patterns); PKA (protein kinase A); PLA2(phospholipase A2); POX (the peroxidase or peroxide reductase component of COX & putative target for acetaminophen); TRPV1 (transient receptor potential subtype V1). Adapted from Chen et al (2013).
Mechanisms Underlying Analgesic Effects (Peripheral & Central)
| Table 1: Molecular Targets for Analgesics | |||
|---|---|---|---|
| Drug Class | Target | Mechanism | Reference |
| Acetaminophen | POX (the overlooked part of “COX”) | Inhibition by Reduction of Fe4+ to Fe3+ | Aronoff et al (2009) |
| Corticosteroids | PLA2 & Gene Transcription | Inhibition of PLA2 & ↓COX2 | Chrousos (2024) |
| NSAIDS | COX 1 & 2 | Inhibition of PGE2 Production / Binds to COX active site | Kafaja et al (2024) |
| Opioids | Kv Channels | β/γ Channel Activation / Hyperpolarization | Schumacher et al (2024) |
| Opioids | Cav2.2 (N-type) | Gi Inhibition / Reduced Ca influx | Schumacher et al (2024) |
| Opioids | HCN2 | Gi mediated ↓cAMP / Reduced Na influx | Ingram (2000) |
| Suzetrigine | Nav1.8 | Na channel (conduction) block | Osteen et al.(2025) |
Different Identified Sites and Cellular Mechanisms of Opioid Induced Analgesia
There is considerable evidence that systemically administered opioid analgesic drugs exert their analgesic effects at multiple sites, including: a) peripheral opioid receptors (Ingram & Williams, 1994; Stein & Lang, 2009), b) ascending pain pathways that include neurotransmission between primary afferent fibers and second order neurons in the dorsal horn of the spinal cord, c) the ‘pain matrix’ areas of the brain where pain messages are integrated (Ossipov et al, 2010); and d) regulation of descending inhibitory pain pathways by mu receptors expressed by neurons in the periacqueductal grey and rostral ventral medulla (Ingram 2000; Fields, 2004; Bingham et al, 2009; Ossipov et al, 2010; Zhang et al 2015). Figures 5, 6 & 7 summarize the major mechanisms by which opioids exert their analgesic effects in the periphery, spinal cord, and descending inhibitory pathways.
Peripheral Mechanism of Opioid Analgesia
-
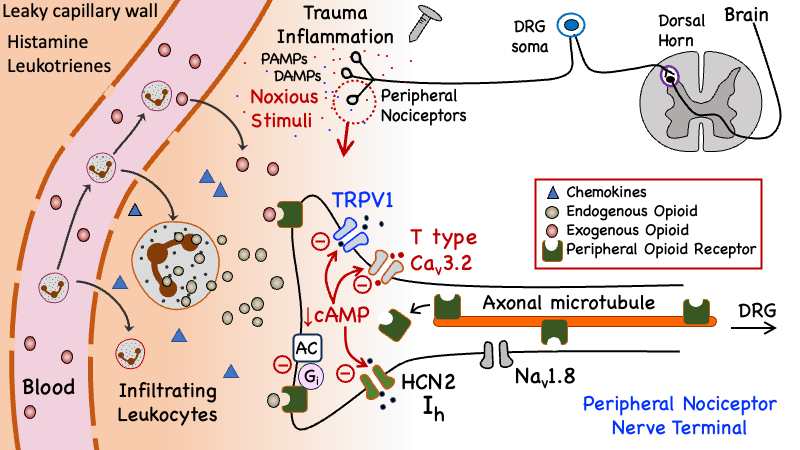
Figure 5. Peripheral mechanism of opioid analgesia. Opioid peptide-containing circulating leukocytes extravasate upon activation of by binding to adhesion molecules (eg. ICAM-1 & beta2 integrin) expressed on the epithelial lining of inflamed blood vessels. In addition, chemokines and norepinephrine released from sympathetic nerve endings can also produce the release of endogenous opioids by activating their respective receptors expressed on the surface of leukocytes. Both exogenous opioids (red circles) and endogenous opioid peptides (light green circles) bind to opioid receptors (MOR, DOR & KOR) that are synthesized in dorsal root ganglia and then transported along intra-axonal microtubules to both peripheral and central terminals of primary afferent sensory neurons. The subsequent inhibition of excitatory ion channels (e.g. TRPV1, Cav3.2, HCN2) results in anti-nociceptive / analgesic effects. Adapted from Stein & Lang (2009) and Iwaszkiewicz et al (2013).
Spinal Cord Mechanism of Opioid Analgesia
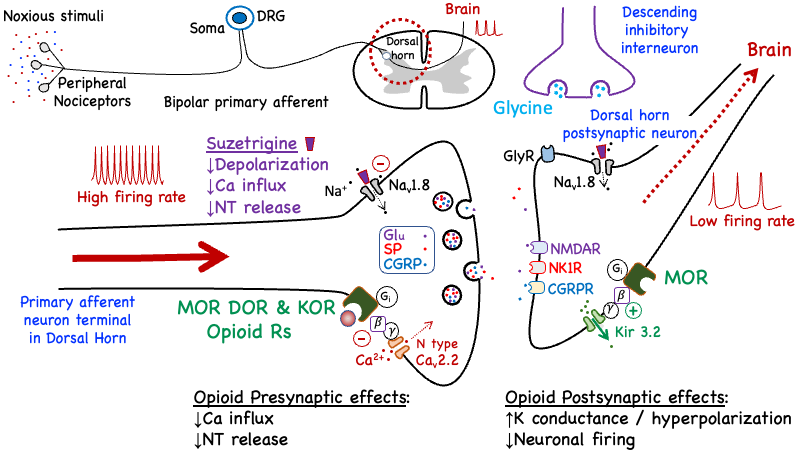
-
Figure 6. Simplified schematic of the primary cellular mechanisms by which suzetrigine and opioid agonists mediate analgesic effects to suppress nociceptive pain at the level of the spinal cord. The top left panel shows a schematic of the neuronal circuit starting with the primary afferent pain fiber that conducts action potentials from the pain receptors located at the site of injury, to the synaptic junction located in the dorsal horn of the spinal cord. The bottom panel shows a magnified view of the synaptic junction identified by the broken red circle in the top panel. Stimulation of opioid receptors (MOR, DOR & KOR) in presynaptic nerve terminal reduces calcium influx and subsequent release of neurotransmitters responsible for transmission of pain signals to the brain. Stimulation of postsynaptic MORs increases K conductance, causing hyperpolarization and a reduction of neuronal firing rate. Suzetrigine is a non-opioid selective blocker of Nav1.8, a TTX-resistant sodium channel isoform expressed in peripheral afferent nociceptive neurons and the dorsal root ganglion, but is not expressed by neurons within the CNS. CGRP: Calcitonin Gene-Related Peptide; CGRPR: Calcitonin Gene-Related Peptide Receptor; DRG: Dorsal Root Ganglion; Glu: Glutamate; GlyR: Glycine Receptor; SP: Substance P; NK1R: Neurokinin 1 (Substance P) Receptor; NMDAR: N-methyl-D-aspartic Acid Receptor.
Glycine (as well as GABA) are known to play an important role as an inhibitory neurotransmitters released by nerve terminals of interneurons that are part of the descending modulatory pathway. When activated, these inhibitory interneurons dampen pain signals traveling up the spinal cord to the brain, producing an antinociceptive effect (Gallagher et al 2022; Liu & Kelliher 2022). As illustrated in Figure 7, this descending modulatory pathway has been identified as an important site of action for the analgesic effects of opioid analgesics, in addition to modulation of the neurotransmission of pain signals in the dorsal horn (Figure 5) and periphery (Figure 6).
Opioid Analgesic Mechanisms in the Brain Stem
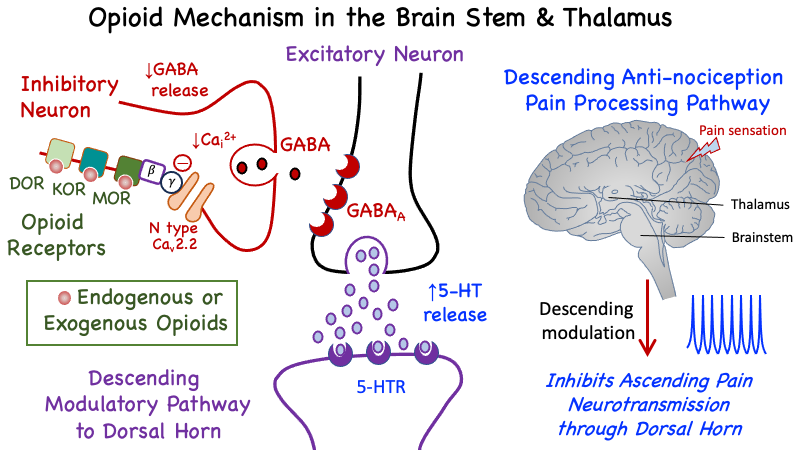
-
Figure 7. Effects of opioids on pain neurotransmission in the Brain. See Figure 3 for additional details on the role of the descending modulatory pathway in pain processing and neurotransmission.
Why Acetaminophen and NSAIDs are combined with Opioids
The combination of NSAIDs (aspirin) or acetaminophen with opioids has been found to produce an analgesic effect that is either additive or synergistic in studies in animal models of pain, and in clinical practice (Curatolo & Sveticid, 1002; Ortiz & Castaneda-Hernandez, 2008). A list of such drug combinations is provided in Table 2. Such drug combinations are associated with a lower incidence of dose-dependent side effects, and in some situations a superior analgesic effect than provided by either drug alone, as well as the ability to control pain and provide adquate analgesia with the use of lower doses of opioids.
| Table 2: FDA Approved Analgesic Combinations | |
|---|---|
| Drugs | Trade Name |
| Acetaminophen + Codeine | Tylenol-Codeine ® |
| Acetaminophen + Hydrocodone | Vicodin® |
| Acetaminophen + Oxycodone | Percocet ® |
| Acetaminophen + Tramadol | Ultracet ® |
| Asprin + Hydrocodone | Percodan ® |
While the ability of COX inhibitors to interfere with the synthesis of PGE2, which plays an important role in nociception in both peripheral nerve endings (Fig 4) and pain neurotransmission at the level of the spinal cord (Fig 5) no doubt contributes to their analgesic effects when combined with mu-receptor agonists, evidence for an additional central mechanism has been discovered that likely contributes to the synergistic outcome often observed when either acetaminophen or an NSAID (e.g. aspirin) is combined with an opioid such as codeine, hydrocodone, oxycodone or the opioid-like drug tramadol (Vaughan et al, 1997). As illustrated in a reproduction of observations first published by Vaughan et al (1997), addition of the NSAID (COX inhibitor) indomethacin can produce a 3-fold shift of the IC50 (potency) of the mu agonist DAMGO from 96 to 32 nM (see Fig 3b in Vaughan et al, 1997). A similar interaction was also observed between morphine and indomethacin, where 10 uM morphine + 10 uM indomethacin reduced electrically stimulated IPSCs by 54 ± 8% (n=5), compared to 24 ± 6% (n=8) with 10 uM morphine alone. In this study, Vaughn and coworkers provided evidence for a novel mechanism whereby mu-opioid agonists stimulate the release of arachidonic acid by a Gi-mediated activation of phospholipase A2, and inhibitors of either COX or 5-LOX enhance the effect of opioids by inducing a shift of arachidonic acid into the 12-LOX pathway, resulting in enhanced production of 12-HPETE metabolites, which are known activators of voltage dependent K channels (Vaughan et al, 1997; Ingram 2000).
-
Experimental evidence supporting this model includes:
-
mu-opioid agonists open an inwardly rectifying K currents in neurons of the mid-brain periqueductal grey (Christie et al, 2000).
-
the effect of morphine on IPSCs is enhanced in the presence of the 5-lipoxygenase inhibitor caffeic acid (Vaughan et al, 1997)
-
mu-opioid mediated presynaptic inhibition of GABAergic neurotransmission is abolished by inhibitors of phospholipase A2 and specific 12-LOX inhibitors, whereas inhibitors of COX and 5-LOX enhance the inhibitory effect of mu opioid agonists on GABA release (Christie et al, 2000).
-
the effects of mu-opioid agonists on GABAergic transmission is mimicked by both arachidonic acid and 12-LOX metabolites (Christie et al, 2000).
-
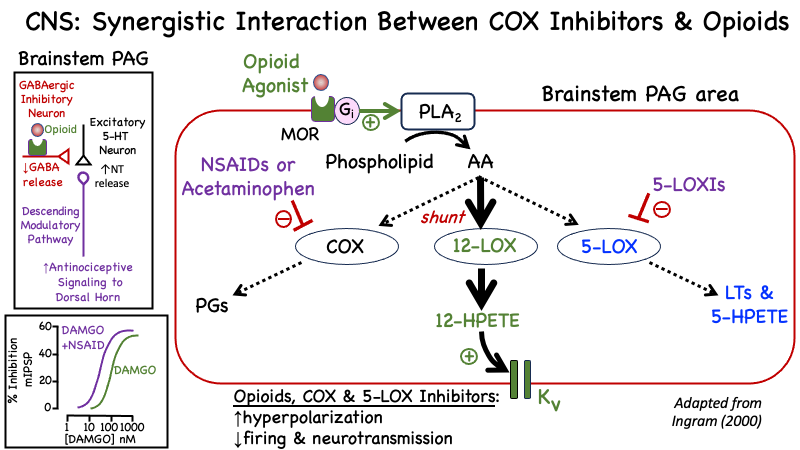
-
Figure 8. A proposed cellular mechanism to explain how NSAIDs/Acetaminophen and opioids exert synergistic analgesic effects in the CNS. Adapted from Ingram & Williams (1999) & Ingram SL(2000). Opioids are known to stimulate PLA2 via a Gi mediated pathway, resulting in increased formation of arachidonic acid. Arachidonic acid serves as a substrate for both COX, 12-LOX and 5-LOX. When the COX pathway is inhibited by an NSAID or acetaminophen, more of AA becomes shunted down the 12-LOX alternative pathway, resulting in increased levels of 12-HETE, a fatty acid derivative of AA that activates voltage dependent potassium channels. Top left panel: illustrates a schematic of the major components of the circuitry involved in regulating the serotonergic neurons of the descending modulatory pathway that excite inhibitory GABA or Glycine interneurons in the dorsal horn, which when activated inhibit neurotransmission in the ascending nociceptive pathway that normally sends pain signals up the spinal cord to numerous regions of the CNS, including the thalamus and cerebral cortex. Lower left panel: illustrates the effect of adding 10 uM indomethacin (an NSAID) to the concentration response relationship for the mu opioid agonist DAMGO on GABA-mediated formation of mIPSCs in the periaqueductal gray region of the brainstem. See Vaughan et al (1997) or Ingram (2000) for the actual raw data. Abbreviations: AA: Arachidonic acid; COX: Cyclooxygenase; DAMGO: a synthetic enkephalin analog & selective MOR receptor agonist; Kv: voltage dependent potassium channel; PG: Prostaglandins; MOR: mu-opioid receptor; NSAID: Non Steroidal Anti-Inflammatory Drug (e.g. ibuprofen, indomethacin or aspirin); 5-LOX: Arachidonate 5-lipoxygenase; 5-LOXIs: inhibitors of 5-LOX (e.g. zileuton); 12-HPETE: 2-Hydroxyeicosatetraenoic acid; 12-LOX: arachidonate 12-lipoxygenase. Adapted from Ingram & Williams (1994) and Christie et al (1999).
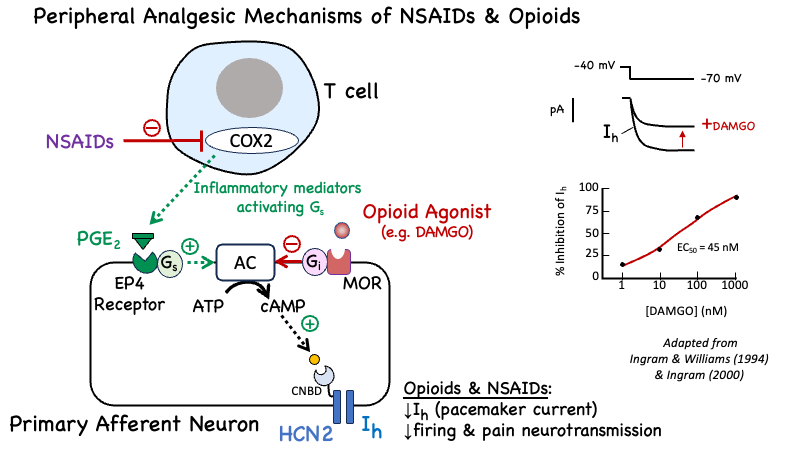
-
Figure 9. A mechanism for potential synergistic interaction between opioids and NSAIDs to reduce the firing of HCN2 ion channels which have been identified as “pacemakers of pain” (Emery et al, 2012). NSIAD-induced inhibition of upregulated COX2 in activated leukocytes will reduce the production of PGE2. This will reduce the stimulation of EP4 prostanoid receptors that are expressed on peripheral nerve endings, and are coupled to adenylyl cyclase (AC) via Gs. Activation of mu opioid receptors (MOR) will inhibit AC via Gi. Both mechanisms will result in reduced intracellular cAMP levels, and reduce the activation of the cyclic nucleotide modulated cation channel (HCN2), resulting in a reduced pacemaker current (Ih and a reduced rate of pacemaker-induced production of action potentials that would be conducted to the dorsal horn through the superior cerivical ganglion. The schematic shown on the left was adopted from Ingram (2000), and the illustration of the effect of the selective MOR agonist DAMGO is adapted from the original data published by Ingram & Williams (1994).
References
-
Anstie FE (1865): The Definition of Narcosis. Chapter IV. In: Stimulants and Narcotics: Their Mutual Relations: With Special Researches on the Action of Alcohol, Aether and Chloroform on the Vital Organism. Lindsay and Blakiston, Editors. Pg 152.
-
Aronoff DM et al (2009): New insights into the mechanism of action of acetaminophen: Its clinical pharmacologic characteristics reflect its inhibition of the two prostaglandin H2 synthases. Clinical Pharmacology & Therapeutics. 79:9-19. DOI: 10.1016/j.clpt.2005.09.009
-
Beaver WT (1984): Combination Analgesics. Am J Med 77(3):38-53. PMID: 6486130 DOI: 10.1016/s0002-9343(84)80101-1
-
Benitez-Angeles M et al (2020): TRPV1: Structure, Endogenous Agonists, and Mechanisms. Int Journal Molecular Sciences. 21(10):3421. doi: 10.3390/ijms21103421 PMID: 32408609
-
Bingham B et al (2009): The molecular basis of pain and its clinical implications in rheumatology. Nat Clin Pract Rheumatol. 5(1):28-37. doi: 10.1038/ncprheum0972. PMID: 19098926
-
Brown DA, Yule DI (2007): Protein kinase C regulation of P2X3 receptors is unlikely to involve direct receptor phosphorylation. Biochim Biophys Acta 1773(2):166-75. doi: 10.1016/j.bbamcr.2006.09.020.
-
Butour JL, et al (1997): Recognition and activation of the opioid receptor-like ORL 1 receptor by nociceptin, nociceptin analogs and opioids. Eur J of Pharmacol. 321(1):97–103. doi:10.1016/S0014-2999(96)00919-3. PMID 9083791.
-
Chen L et al (2013): Prostanoids and inflammatory pain. Prostaglandins & other Lipid Mediators. 104–105 (2013) 58– 66. http://dx.doi.org/10.1016/j.prostaglandins.2012.08.006
-
Christie MJ et al (1999): Opioids, NSAIDs and 5-lipoxygenase inhibitors act synergistically in brain via arachidonic acid metabolism. Inflamm Res 48 (1999) 1–4. DOI: https://doi.org/10.1007/s000110050367
-
Christie MJ et al (2000): Cellular Actions Of Opioids And Other Analgesics: Implications For Synergism In Pain Relief. Clinical and Experimental Pharm Physiol 27:520-523. https://doi.org/10.1046/j.1440-1681.2000.03291.x
-
Chrousos GP (2024): Adrenocorticosteroids & Adrenocortical Antagonists (Chapter 39). In: Katzung’s Basic and Clinical Pharmacology. 16th Ed. Vanderah TW (Editor); McGraw-Hill (Access Medicine).
-
Curatolo M, Sveticic G (2002): Drug combinations in pain treatment: a review of the published evidence and a method for finding the optimal combination. Best Pract Res Clin Anaesthesiol 16:507–19. doi: 10.1053/bean.2002.0254. PMID: 12516888
-
Dalle C et al (2005): Peripheral Block of the Hyperpolarization-Activated Cation Current (Ih) Reduces Mechanical Allodynia in Animal Models of Postoperative and Neuropathic Pain. Regional Anesthesia and Pain Medicine. 30(3):243–248. doi:10.1016/j.rapm.2005.01.010
-
Dhaliwal A, Gupta M (2023): Physiology, Opioid Receptor. [Updated 2023 Jul 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK546642/
-
Dray A (1995): Inflammatory mediators of pain. Br J Anaesthesia 75:125–131
-
Emery EC et al (2011): HCN2 ion channels play a central role in inflammatory and neuropathic pain. Science 333(6048):1462-6. doi: 10.1126/science.1206243. PMID: 21903816
-
Emery EC et al (2012): HCN2 ion channels: an emerging role as the pacemakers of pain. Trends Pharmacol Sci 33(8):456-63. doi: 10.1016/j.tips.2012.04.004. PMID: 22613784
-
Fields H (2004): State-dependent opioid control of pain. Nature Reviews Neuroscience. 5(7):565-575. DOI: 10.1038/nrn1431; PMID: 15208698
-
Gallagher C et al (2022): Positive Allosteric Modulators of Glycine Receptors and Their Potential Use in Pain Therapies. Pharmacol Rev 74(4):933–961. doi: 10.1124/pharmrev.122.000583 PMID: 36779343
-
Harvey RJ et al (2004): GlyR 3: An Essential Target for Spinal PGE2-Mediated Inflammatory Pain Sensitization. Science. 304(5672):884-887. DOI: https://doi.org/10.1126/science.1094925
-
Hermann S et al (2017): Protein kinase A regulates inflammatory pain sensitization by modulating HCN2 channel activity in nociceptive sensory neurons. Pain 158 (2017) 2012–2024. http://dx.doi.org/10.1097/j.pain.0000000000001005
-
Huang CL et al (1995): Evidence That Direct Binding of to the GIRK1 Gβγ Protein-Gated Inwardly Rectifying K+ Channel Is Important for Channel Activation. Neuron 15:1133-1143. PMID: 7576656 DOI: 10.1016/0896-6273(95)90101-9
-
Ingram SL (2000): Cellular and molecular mechanisms of opioid action. Prog Brain Res 129:483-92. doi: 10.1016/S0079-6123(00)29035-3. PMID: 11098712
-
Ingram SL, Williams JT (1994): Opioid Inhibition of lh via Adenylyl Cyclase. Neuron 13:179-186. DOI: 10.1016/0896-6273(94)90468-5 PMID: 7519024
-
Iwaszkiewicz KS et al (2013): Targeting peripheral opioid receptors to promote analgesic and anti-inflammatory actions. Front Pharmacol 4(2013): https://doi.org/10.3389/fphar.2013.00132
-
Kafaja TK et al (2024): Nonsteroidal Anti-Inflammatory Drugs, Disease-Modifying Antirheumatic Drugs, Nonopioid Analgesics, & Drugs Used in Gout (Chapter 36). In: Katzung’s Basic and Clinical Pharmacology. 16th Ed. Vanderah TW (Editor); McGraw-Hill (Access Medicine).
-
Liu S, Kelliher L (2022): Physiology of pain—a narrative review on the pain pathway and its application in the pain management. Digestive Medicine Research 5:56 | https://dx.doi.org/10.21037/dmr-21-100
-
Ortiz MI, Castaneda-Hernández G (2008):Examination of the interaction between peripheral lumiracoxib and opioids on the 1% formalin test in rats. Eur J Pain 12:233-241. doi: 10.1016/j.ejpain.2007.05.008.
-
Ossipov MH et al (2010): Central modulation of pain. J Clin Invest 120:3779-3787. doi:10.1172/JCI43766. PubMed: 21041960.
-
Osteen, JD et al.(2025): Pharmacology and Mechanism of Action of Suzetrigine, a Potent and Selective NaV1.8 Pain Signal Inhibitor for the Treatment of Moderate to Severe Pain. Pain and Therapy. doi:10.1007/s40122-024-00697-0. PMID 39775738.
-
Paukert M et al (2001): Inflammatory mediators potentiate ATP-gated channels through the P2X(3) subunit. J Biol Chem 276(24):21077-82. doi: 10.1074/jbc.M101465200. PMID: 11264291
-
Prescott SA, Ratté S (2017): Somatosensation and pain, In: Conn’s Translational Neuroscience. P. M. Conn (Editor). Academic Press, San Diego, CA. PP: 517–539.
-
Raingo J et al (2007): Alternative splicing controls G protein–dependent inhibition of N-type calcium channels in nociceptors. Nat Neurosci 10:285–292. https://doi.org/10.1038/nn1848
-
Saleem MZ, Nisar MA (2022): Mechanism of opioids action and their receptor-dependent signaling. Chapter 23 In:How Synthetic Drugs Work. Insights into Molecular Pharmacology of Classic and New Pharmaceuticals. 1st Edition.Kazmi I, Karmakar S et al (Editors). Elsevier eBook ISBN: 978032391368. PP:529-544.
-
Scholz A (2007): Nav1.8 Voltage-Gated Sodium Channel. In: xPharm: The Comprehensive Pharmacology Reference. Pages:1-8. Elsevier. https://doi.org/10.1016/B978-008055232-3.60414-3
-
Schumacher MA et al (2024): Opioid Agonists & Antagonists (Chapter 31). In: Katzung’s Basic and Clinical Pharmacology. 16th Ed. Vanderah TW (Editor); McGraw-Hill (Access Medicine).
-
Stein C, Lang LJ (2009): Peripheral mechanisms of opioid analgesia. Curr Opinion Pharmacol 9:3-8. DOI 10.1016/j.coph.2008.12.009
-
Vaughan CW et al (1997): How opioids inhibit GABA-mediated neurotransmission. Nature 390(6660):611-4. DOI https://doi.org/10.1038/37610 PMID: 9403690
-
Wirkner K et al (2005): Regulation of Human Recombinant P2X3 Receptors by Ecto-Protein Kinase C. Journal of Neuroscience 25(34):7734-7742. https://doi.org/10.1523/JNEUROSCI.2028-05.2005
-
Wood TE, Goldberg MB (2022): Chapter 120: Molecular Mechanisms of Microbial Pathogenesis. In: Harrison’s Principles of Internal Medicine, 21e. Loscalzo J, Fauci A et al (Editors). McGraw Hill.
-
Zhang W et al (2015): DAMGO depresses inhibitory synaptic transmission via different downstream pathways of mu opioid receptors in ventral tegmental area and periaqueductal gray. Neuroscience. 301:144-54. http://dx.doi.org/10.1016/j.neuroscience.2015.05.077
本文来自pharmwiki,本文观点不代表OUQ技术分享立场,转载请联系原作者。

 微信打赏,为服务器增加50M流量
微信打赏,为服务器增加50M流量  支付宝打赏,为服务器增加50M流量
支付宝打赏,为服务器增加50M流量